Laboratory findings may vary according to the degree of hepatotoxicity. See TABLE 1 for a summary of these stages. The clinical manifestations of acetaminophen toxicity are typically divided into four stages. This makes the time of ingestion, the quantity, and the formulation of acetaminophen ingested extremely important for diagnosis of liver toxicity. 5 Clinical Presentation and Laboratory Findingsīecause clinical symptoms of end-organ toxicity do not manifest until 24 to 48 hours after acute ingestion, most patients with acetaminophen overdose will be asymptomatic initially. 5 Children who ingest >350 mg/kg and are not appropriately treated are at high risk for severe hepatotoxicity. In healthy children aged 1 to 6 years, the minimum toxic dose of acetaminophen as a single ingestion is 150 mg/kg, and acute ingestion of ≥250 mg/kg poses a significant risk for acetaminophen-induced hepatotoxicity. 3,5 Standard and Toxic Acetaminophen Dosagesįor children aged 150 mg/kg or 12 g of acetaminophen in adults is considered a toxic dose and carries a high risk of liver damage. For this reason, the FDA advised in August 2013 that anyone who has a skin reaction, such as the development of a rash or blister, while taking acetaminophen should stop using the drug and seek immediate medical care. Research shows that acetaminophen may also cause potentially fatal skin disorders, such as Stevens-Johnson syndrome, toxic epidermal necrolysis, and acute generalized exanthematous pustulosis. In children aged 325 mg of acetaminophen per tablet, capsule, or other dosage form no longer be prescribed because of the risk of liver damage. Acetaminophen is rapidly and completely absorbed from the gastrointestinal (GI) tract.Īcetaminophen overdose can occur at any age. This is partly due to the contraindication of aspirin in pediatric patients (Reye syndrome). 3Īcetaminophen is one of the most commonly used medications for pain and fever reduction in children. 
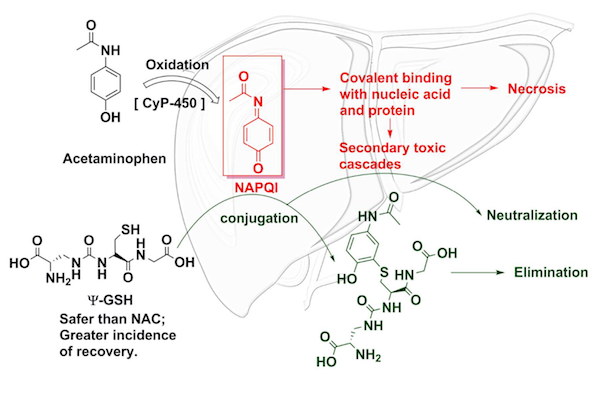
Combination formulations such as hydrocodone-acetaminophen and oxycodone-acetaminophen are considered controlled substances and require a prescription. In the U.S., >25 billion doses are sold yearly in the form of 325-mg and 500-mg immediate-release tablets and 650-mg extended-release products marketed for the treatment of arthritis. Acetaminophen is available in a variety of forms, such as elixirs, suspensions, tablets (dissolvable and chewable), caplets, capsules, and paraffin-base rectal suppositories. 1Īcetaminophen, or N-acetyl- p-aminophenol (APAP), is available in more than 200 OTC and prescription medications under different brand or trade names. for inpatient adults and children aged >2 years. In 2011, an IV formulation of acetaminophen was approved in the U.S. market in 1947, it was available only by prescription. When acetaminophen was introduced to the U.S. This article will briefly review the history, safety profile, clinical presentation, and treatment of acetaminophen intoxication. Acetaminophen exposure alone resulted in 65 deaths, and acetaminophen combinations resulted in 42 deaths. The 2014 annual report of the American Association of Poison Control Centers’ National Poison Data System noted 50,396 single exposures to acetaminophen alone and 22,951 single exposures to acetaminophen in combination with other drugs. Additionally, with extended-release pills, acetaminophen remains in the body for a longer period of time. These include sleeping pills and medications for the common cold, flu, and allergies. Accidental overdose may occur when a person takes too much acetaminophen because the pain or fever does not diminish after the recommended dose, or when a person takes too many different medications containing acetaminophen. 1 For most people, it is safe to take 4,000 mg (4 g) of acetaminophen within a 24-hour period. The American Association of Poison Control Centers calls acetaminophen one of the most common pharmaceuticals associated with both intentional and unintentional poisoning and toxicity. 1Īcetaminophen overdose is also known as acetaminophen poisoning. In the United States, acetaminophen toxicity is the most common cause of acute liver failure and the second most common cause of liver failure requiring transplantation. This is because acetaminophen is metabolized in the liver. If used in proper therapeutic doses, acetaminophen has an excellent safety profile however, its major side effect is hepatotoxicity, which can occur after overdose or misuse. Unlike typical nonsteroidal anti-inflammatory drugs, acetaminophen has only a minor effect on tissue cyclooxygenase activity and appears to produce analgesia by increasing pain thresholds, perhaps through inhibition of the nitric oxide pathway that is activated by many pain neurotransmitter receptors. 
Acetaminophen (known as paracetamol in Europe) is one of the most commonly used oral medications for pain and fever reduction. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
